
Chylous cardiac tamponade after single-port VATS lobectomy in NSCLC: case report
Introduction
Chylothorax is a rare complication of cardiothoracic surgery, and the cause is direct injury of the thoracic duct during surgery. Its incidence is higher with cardiac surgery than with thoracic surgery (1). Esophagectomy is the thoracic procedure most frequently associated with postoperative chylothorax (2). The treatment used for chylothorax depends on the amount of chyle in the thorax. However, traumatic chylous pericardial effusion is rare and even more dangerous than chylothorax because it may lead to cardiac tamponade. Mediastinal node dissection, while essential, can potentially damage the thoracic duct or pericardium in lung cancer surgery, although this is a rare occurrence. We experienced a rare case of chylothorax with cardiac tamponade that required pericardiocentesis and thoracic duct ligation. Herein, we present this case of chylous cardiac tamponade after lobectomy with mediastinal lymph node (LN) dissection in non-small cell lung cancer. We present the case in accordance with the CARE Guidelines.
Case presentation
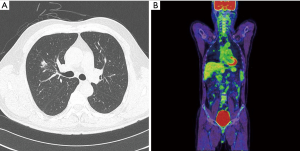
A 54-year-old man was referred to our hospital for a growing mass in the right upper lobe (RUL). He was a nonsmoker, and he had no relevant personal or family medical history. Mixed ground-glass opacity (GGO), measuring approximately 1.8 cm, was identified by computed tomography (CT). The mass showed peripheral spiculation with a 1.4 cm solid portion in the central lesion of the RUL (Figure 1A). Primary lung cancer was suspected. Stage evaluation was performed, which included brain magnetic resonance imaging (MRI), bronchoscopy, and pulmonary function tests. According to positron emission tomography (PET), the maximum standardized uptake value (SUVmax) was 2.38 (Figure 1B). The clinical stage was T1bN0M0. Because of the central location of the mass, lobectomy was performed without tissue confirmation.
Operative technique
Under general anesthesia, the position of the patient was changed to the left lateral decubitus position. A skin incision 3–4 cm in length was made at the fifth intercostal space. The first step was dissection of the interlobar area to identify the arteries, including the posterior segmental artery of the RUL. The next step was the division of both sides of the fissures of the RUL before vessel division. The reasons for dividing both fissures before vessel division were to obtain a clear view of the vessel anatomy, to achieve the requirement of limited blunt dissection during vessel division, and to decrease the risk of bleeding. The anterior trunk was dissected starting from the azygos vein, and this artery was isolated and divided. Then, the vein of the RUL was dissected from the main pulmonary artery under thoracoscopic view, and the vein was easily dissected from the artery. After division of the vein, the posterior segmental artery was identified and divided. Finally, the bronchus of the RUL was divided after the removal of peribronchial LNs. Inferior ligament division was performed for upward lifting of the remaining lung tissue. Subcarinal LNs were removed, and upper mediastinal LNs were also removed (Video 1).
During upper mediastinal LN dissection (Video 1; 2 minutes and 20 seconds), abnormal serous fluid was identified in the upper mediastinal dissection area. At that time, we believed that the fluid had originated from the pericardial space, as it was the pericardium that was opened during LN dissection; we never suspected the fluid to be chyle.
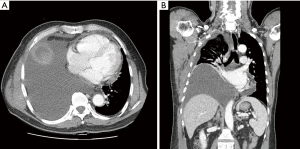
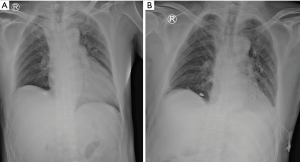
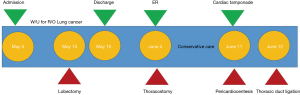
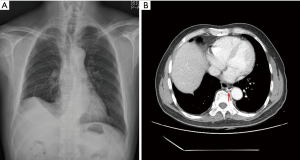
The patient was discharged on postoperative day 4 without any complications. He returned 1 week later after experiencing oozing from the removal site of the chest tube. Serous fluid was drained out, and the wound was closed. After 2 weeks, the patient was referred to the emergency department with mild dyspnea. Chest radiography showed an accumulation of fluid in the right pleural space. Chest CT revealed substantial pleural effusion with mild pericardial effusion (Figure 2). Closed thoracostomy was performed. Two liters of typical milky fluid were drained. Pleural fluid analysis was carried out, and the triglyceride level was 1,213 mg/dL. Thoracic duct ligation was recommended due to the large amount of chylothorax; however, the patient refused the operation. Conservative care was administered, and echocardiography was conducted. According to echocardiography, there was mild pericardial effusion with no functional abnormalities in cardiac function. On day 5 after readmission, because the daily drainage amount exceeded 500 cc, we again recommended surgery, and this time the patient agreed to the operation. However, that night, the patient experienced sudden hypotension (systolic pressure 70 mmHg) with severe agitation and sweating. Emergency echocardiography was performed, and cardiac tamponade was confirmed (Figure 3A). Emergency pericardiocentesis was performed, and 2 liters of pericardial effusion were drained (Figure 3B). The patient’s condition stabilized and he no longer experienced symptoms. Reoperation was performed the next day. Pericardial window and thoracic duct ligation were carried out (Video 2). Severe pleural adhesions were noted between the lower paratracheal region and the right lung that had induced cardiac tamponade. The patient was discharged on postoperative day 5 without any complications (Figure 4).
After 4 months, there was no fluid accumulation in the patient’s thorax or pericardium (Figure 5).
All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review by the editorial office of this journal.
Discussion
Chylothorax is a rare complication that occurs after thoracic surgery, with an incidence of approximately 2%. Postoperative chylothorax has been reported to occur after various types of cardiothoracic surgery, including esophagectomy, mediastinal mass resection, and heart surgery. Chylothorax may occur after lung cancer surgery, and it most commonly accompanies mediastinal LN dissection (3). Postoperative chylothorax is caused by thoracic duct injury, which goes unrecognized by surgeons during surgery in the majority of cases.
Postoperative chylothorax is a potentially dangerous condition if left untreated. However, most clinical cases are not urgent. Prolonged chyle leakage may lead to malnutrition and sepsis. If chylothorax is diagnosed by pleural fluid analysis, most cases of postoperative chylothorax can be successfully managed with conservative care or thoracic duct ligation (3). The management of chylothorax has been well described in previous literature (4).
However, chylous cardiac tamponade is a life-threatening disease, and the clinical course may be urgent (5). Cardiac tamponade is a rare complication after lobectomy (6). The cause of cardiac tamponade is pericardial injury during surgery (7); it may be associated with either direct or traction injury. However, holes in the pericardium usually do not result in severe problems because there is usually communication between the pericardial space and the pleural space; thus, the presence of cardiac tamponade after lobectomy is considered to be uncommon. However, chylous cardiac tamponade may progress along a more severe clinical course should chyle leakage persist and the chyle accumulate in the pericardial space if a pericardial hole is present. Furthermore, drainage is important for the treatment of chyle leakage. There is no effective drainage method for chylous pericardial effusion after thoracic surgery.
In this case, uniportal lobectomy was performed. It may be argued that uniportal lobectomy introduces more complications than conventional thoracoscopic lobectomy. However, many thoracic surgeons have demonstrated that there is no difference between the two regarding complications (8). In our experience, no previous cases of chylothorax had been observed with the use of a uniportal procedure. We believe that this eventful condition arises from anatomical variations of the thoracic duct.
We identified pericardial injury and fluid accumulation in the lower paratracheal region during LN dissection. However, one limitation of this case is that we never recognized that the accumulating fluid was chyle, and the opening of the pericardium was not small, so we thought that the pericardial space was easily communicating with the pleural space. Later, as pleural adhesion progressed, the route of communication between the pericardial and pleural spaces was obstructed, and cardiac tamponade developed.
Conclusions
An initial surgical procedure is recommended when the amount of chest tube drainage is 1 liter or more after thoracostomy (3). In addition, as pericardial injury can lead to serious complications, we recommend thoracic duct ligation when chylothorax is present with pericardial effusion on imaging studies. If there is abnormal fluid collection during mediastinal LN dissection with the pericardial opening during thoracic surgery, pericardial closure or pericardial window should be recommended at the time of surgery. If chyle leakage is suspected, it is best to consider thoracic duct ligation.
Acknowledgments
Funding: None.
Footnote
Conflicts of Interest: Both authors have completed the ICMJE uniform disclosure form (available at https://ccts.amegroups.com/article/view/10.21037/ccts.2020.03.03/coif). HWJ serves as an unpaid editorial board member of Current Challenges in Thoracic Surgery from January 2020 to December 2021. The other author has no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. All procedures performed in this study were in accordance with the ethical standards of the institutional and/or national research committee(s) and with the Helsinki Declaration (as revised in 2013). Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review by the editorial office of this journal.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Zaidenstein R, Cohen N, Dishi V, et al. Chylothorax following median sternotomy. Clin Cardiol 1996;19:910-2. [Crossref] [PubMed]
- Fujita T, Sato T, Sato K, et al. Clinical manifestation, risk factors and managements for postoperative chylothorax after thoracic esophagectomy. J Thorac Dis 2019;11:S198-201. [Crossref] [PubMed]
- Reisenauer JS, Puig CA, Reisenauer CJ, et al. Treatment of Postsurgical Chylothorax. Ann Thorac Surg 2018;105:254-62. [Crossref] [PubMed]
- Bender B, Murthy V, Chamberlain RS. The changing management of chylothorax in the modern era. Eur J Cardiothorac Surg 2016;49:18-24. [Crossref] [PubMed]
- Alsmady MM, Aladaileh MA, Al-Zaben K, et al. Chylopericardium presenting as cardiac tamponade secondary to mediastinal lymphangioma. Ann R Coll Surg Engl 2016;98:e154-6. [Crossref] [PubMed]
- Pillai JB, Barnard S. Cardiac tamponade: a rare complication after pulmonary lobectomy. Interact Cardiovasc Thorac Surg 2003;2:657-9. [Crossref] [PubMed]
- Kosugi SI, Hashimoto T, Sato Y, et al. Cardiac tamponade communicating with a posterior mediastinal chylocele after esophagectomy. J Surg Case Rep 2017;2017:rjx216. [Crossref] [PubMed]
- Ji C, Xiang Y, Pagliarulo V, et al. A multi-center retrospective study of single-port versus multi-port video-assisted thoracoscopic lobectomy and anatomic segmentectomy. J Thorac Dis 2017;9:3711-8. [Crossref] [PubMed]
Cite this article as: Jeon HW, Kim YD. Chylous cardiac tamponade after single-port VATS lobectomy in NSCLC: case report. Curr Chall Thorac Surg 2020;2:41.


