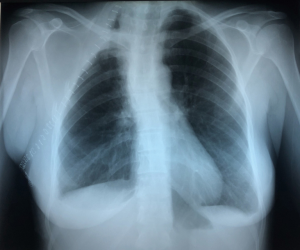
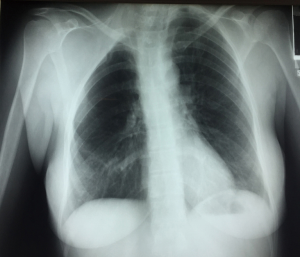
A patient with a localized malignant pleural mesothelioma and 15-year disease-free survival
Introduction
Malignant pleural mesothelioma (MPM) is a rare neoplasm that arises from the pleura, although it is its most common primary malignancy. Prognosis is poor, with a median survival that is usually under 12 months and a 5-year survival rate under 5% (1). Localized malignant pleural mesothelioma (LMPM) is an even rarer malignancy of the pleura that the World Health Organization clearly distinguished from MPM in 2004 (2). LMPM has the same histo-pathological features as MPM, but it can be differentiated from it because it does not spread along the pleura (3). The same histological subtypes have been identified in LMPM, being the most frequent the epithelioid type (1,4,5). In diffuse MPM the epithelioid type has better prognosis than the sarcomatous or biphasic types, but this has not been proved in LMPM (6).
Case presentation
A 40-year-old female, smoker of 30 cig/day and no other past medical history, presented with right scapular pain, neck swelling, shortness of breath and signs of neck venous congestion of three-month duration. The patient was transferred from another hospital, where she had had a chest X-ray (Figure 1) showing a mass in the right upper one third of the chest. On admission to our hospital, the patient referred that for the previous three weeks she had had non-purulent expectoration, fever, cold sweat, shortness of breath and decreased sensitivity along the inner aspect of her right arm. Physical examination revealed swelling in the cervical area and ingurgitation of the neck veins.
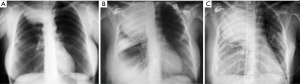
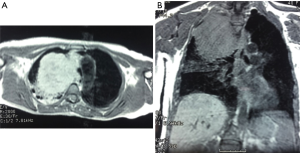
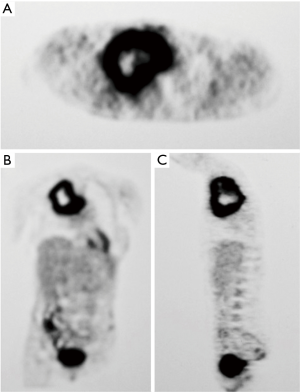
A computed tomography (CT) of the chest revealed an 8 cm × 10 cm mass on the upper part of the right chest, in contact with the posterior chest wall and the upper mediastinum, which was displaced to the contralateral side (Figure 2). A magnetic resonance imaging (MRI) confirmed the CT findings and showed a heterogeneous mass that pushed the mediastinum, with severe tracheal compression but without clear signs of invasion (Figure 3A,B). Pulmonary function tests showed an obstructive pattern, with forced vital capacity (FCV) 88%, forced expiratory volume in 1 second (FEV1) 73% and FCV/FEV1 65%. Complete blood count and routine blood tests were normal, except for an increased LDH. Positron emission tomography (PET) showed an intense uptake in the periphery of the mass and in the upper medial cervical line, although it was impossible to differentiate whether it was an enlarged lymph node or a lesion of the right vocal cord (Figure 4A,B,C). Fiberoptic bronchoscopy showed signs of extrinsic tracheal compression, but yielded no diagnosis. A video-mediastinoscopy indicated to diagnose and stage the tumour allowed the taking of biopsies from the tumour and from enlarged right paratracheal lymph nodes. A few days after video-mediastinoscopy, the patient suffered from increasing shortness of breath that led to acute respiratory insufficiency, sever hypoxemia and haemodynamic instability that required orotracheal intubation, mechanical ventilation and the administration of vasoactive drugs, respectively (Figure 1B). The patient remained in critical condition for 72 hours.
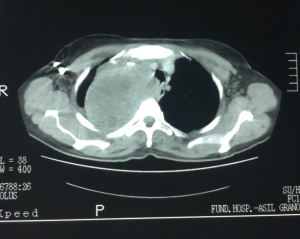
Six days after intubation, when haemodynamic stability had been achieved, the patient underwent postero-lateral thoracotomy, although there was no histo-pathological diagnosis of the mass (Figure 1C). The tumour on the upper part of the chest was found to invade the supero-posterior chest wall and to strongly adhere to the right upper lobe. A right upper lobectomy en bloc with the posterior segments of the 2nd to 4th ribs, including the tissues extending to the paravertebral spaces, and an extrapleural detachment of the tumour at the apex of the pleural cavity and on the mediastinal side were performed, as well as systematic nodal dissection. Two days after surgery, the pathological report informed that the mass biopsied during mediastinoscopy was suggestive of MPM, epithelial subtype.
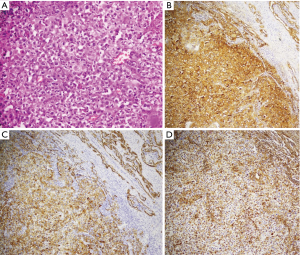
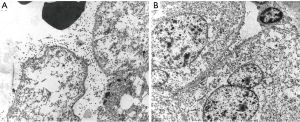
The postoperative course was uneventful and the patient was discharged on the 8th postoperative day (Figure 5). Routine pathological examination of the resected specimen revealed a highly undifferentiated malignant neoplasm with immunohistochemical (IHC) characteristics that could not differentiate between carcinoma and mesothelioma. IHC was positive for adenocarcinoma markers (epithelial membrane antigen and cytokeratin 7) and also for mesothelial markers (vimentin) (Figure 6). All removed lymph nodes were negative. Electron microscopy revealed the tumour was a MPM (Figure 7). The patient received sequential adjuvant chemo-radiotherapy (3 cycles of cisplatinum and gemcitabine, and 63Gy in 35 fractions, 1.8 Gy/fraction). She is disease-free 15 years after operation (Figure 8).
Discussion
Due to its rareness and scarce reports, the natural history of LMPM is not entirely known (4). Therefore, it has been studied in comparison with the diffuse type of MPM. In this regard, the correlation between asbestos exposure and diffuse MPM is widely known, but when it comes to LMPM, although a history of exposure to asbestos might also be present, it is less frequent (4,6), and, therefore, the correlation between asbestos and LMPM has been difficult to prove.
LMPM is more often diagnosed in asymptomatic patients that get a chest X-ray for another reason, and, differently from its diffuse counterpart, it less frequently causes respiratory symptoms or pleural effusion in early stages (4). Symptoms are more frequent in advanced stages, where patients can experience dyspnoea and thoracic pain mostly related to compression of neighbouring structures, such as the patient’s case, who showed mild symptoms for months, until the growth of the tumour caused tracheal compression that led to orotracheal intubation. The typical radiological findings in these stages are pleural effusion, pleural thickening, and involvement of peripheral tissues, lung or ribs (1). A case presenting with pulmonary parenchyma involvement without pleural radiologic findings has also been reported (7).
Leaving aside the clinical features the patients might be presenting, when pleural abnormalities are suspected, the initial approach should be based on a chest CT, which provides information about the localization of the tumour and the involvement of surrounding tissues. MRI provides more detailed information about mediastinal involvement, but has a lower sensitivity for the assessment of metastases. Combined PET-TC seems especially effective in the pre-surgical staging of MPM, in estimating treatment response as well as identifying disease relapse, but there is little experience on its value in LMPM. In the case reported herein, the high uptake in the periphery of the tumour and a much lesser uptake in the centre of the mass suggested a fast-growing malignant tumour with central necrosis. Angiography can be performed to assess vessel involvement (1).
The published evidence seems to show LMPM is a disease with better prognosis than diffuse MPM (3,8). The fact that this is a local neoplasm makes it possible to attempt complete resection (4), but since its biological behaviour is not completely understood, multimodal treatment is usually recommended (5,6). A case like the one reported here seems a good example of it. Because of the rapid growth of the tumour, the patient developed acute respiratory insufficiency due to tracheal compression, and an emergency surgery was performed based on imaging, only. The aggressive nature of the tumour and the final histopathological diagnosis justified the indication of adjuvant local radiotherapy and systemic chemotherapy. The good response to the combined local and systemic treatment and the 15-year survival of this patient support the idea that this is a disease than can be cured if complete resection is achieved.
Regarding the possibility of metastasis, there is no clear data on the biological behaviour of the tumour, but the published reports suggest there is an initially localised phase of the disease followed by a phase where haematogenous dissemination is possible. Metastases to the brain, kidneys and stomach have been reported (5,7,9).
Differential diagnosis of LMPM include diffuse MPM, mesothelial cell hyperplasia, primary neoplasms arising from the lung and involving the pleura, and pleural metastases. An accurate medical history and evaluation of radiologic images is mandatory, but definitive diagnosis requires pathologic examination of biopsies or resected specimens. When standard haematoxylin and eosin stains are not enough for a definitive diagnosis, IHC stains can be very useful in the differential diagnosis. Usually, a panel of IHC stains will have at least two mesothelial markers and two non-mesothelial markers (10,11), and additional markers can be included to identify epithelial tissue from the epithelial and mesenchymal tumours that are considered in the differential diagnosis. In cases where IHC stains are unclear, such as our patient’s case, electron microscopy can help reaching a diagnosis. Nowadays, the diagnostic tests are also based on the study of molecular abnormalities (12), which seem to be helpful in differentiating between benign and malignant tumours.
The solitary fibrous tumour of the pleura (SFTP) also has to be considered in the differential diagnosis. This is a rare primary tumour of the pleura: less than 5% of primary tumours of the pleura are SFTP (13), but it is the most common benign primary tumour of the pleura. It was initially believed to be a benign form of mesothelioma, but it has been proven that it arises from mesenchymal tissue instead of the mesothelium. It is most frequently found in asymptomatic patients (14) that get a chest x-ray for other reasons, but they may cause respiratory symptoms such as coughing, chest pain or shortness of breath (13). Thoracic CT also is the recommended imaging technique for initial assessment. It usually identifies a mass of varying size near the chest wall without actually affecting it, with a heterogeneous consistency and possible calcifications in its interior (15). Despite the radiological assessment, the final diagnosis and its benign or malignant nature can only be assessed by a pathologist, which is why a complete resection of the tumour is recommended (14). The differentiation of SFTP from LMPM will be confirmed with specific IHC stains, in which one of the differences found might be a negative staining for cytokeratin, which is a differential factor from mesothelioma (13).
Conclusions
This case report presents a rare but possible evolution of a patient with a disease that is very little known. Because of its low incidence, its biological behaviour and prognosis are difficult to assess. Therefore, it is important to be aware of this possible diagnosis in our daily practice when a patient with pleural disease is being evaluated, not only to avoid an erroneous diagnosis that would lead to an inadequate therapeutic plan, but also because its differential diagnosis includes a very wide range of diseases, from very malignant ones, such as diffuse MPM that would need a much more aggressive treatment and follow-up, to benign pleural tumours that require very different therapies. It is always preferable to perform a surgical procedure having a clear diagnosis, but this case shows that good results can be obtained when good preoperative assessment is performed; and that even in emergency operations such as the one this patient underwent, performing a complete resection of a malignant tumour results in the best chance of long-term survival.
Acknowledgments
Funding: None.
Footnote
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/ccts.2019.09.02). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Yao W, Yang H, Huang G, et al. Massive localized malignant pleural mesothelioma (LMPM): manifestations on computed tomography in 6 cases. Int J Clin Exp Med 2015;8:18367-74. [PubMed]
- Travis WD, Brambilla E, Müller-Hermelink H, et al. Pathology and Genetics of Tumours of the Lung, Pleura, Thymus and Heart. Lyon: IARC Press, 2004.
- Gelvez-Zapata SM, Gaffney D, Scarci M, et al. What is the survival after surgery for localized malignant pleural mesothelioma? Interac Cardiovasc Thorac Surg 2013;16:533-7. [Crossref] [PubMed]
- Mann S, Khawar S, Moran C, et al. Revisiting localized malignant mesothelioma. Ann Diagn Pathol 2019;39:74-7. [Crossref] [PubMed]
- Zardawi SJ, Li BT, Zauderer MG, et al. Localized malignant pleural mesothelioma with renal metastasis. Ox Med Case Reports 2015:170-2.
- Allen TC, Cagle PT, Churg AM, et al. Localized malignant mesothelioma. Am J Surg Pathol 2005;29:866-73. [Crossref] [PubMed]
- Ertan G, Eren A, Ulus S. Rare presentation of a localised malignant pleural mesothelioma with cranial metastasis. BMJ Case Rep 2016. [Crossref] [PubMed]
- Nakas A, Martin-Ucar AE, Edwards JG, et al. Localised malignant pleural mesothelioma: a separate clinical entity requiring aggressive local surgery. Eur J Cardiothorac Surg 2008;33:303-6. [Crossref] [PubMed]
- Hayashi H, Notohara K, Yoshioka H, et al. Localized Malignant Pleural Mesothelioma Showing a Thoracic Mass and Metastasizing to the Stomach. Intern Med 2010;49:671-5. [Crossref] [PubMed]
- Ordóñez NG. Application of immunohistochemistry in the diagnosis of epithelioid mesothelioma: A review and update. Hum Path 2013;44:1-19. [Crossref] [PubMed]
- Husain AN, Colby TV, Ordóñez NG, et al. Guidelines for pathologic diagnosis of Malignant Mesothelioma: 2017 Update of the consensus statement from the International Mesothelioma Interest Group. Arch Pathol Lab Med 2018;142:89-108. [Crossref] [PubMed]
- Galateau-Salle F, Churg A, Roggli V, et al. The 2015 World Health Organization classification of tumors of the pleura: Advances since the 2004 Classification. J Thorac Oncol 2016;11:142-54. [Crossref] [PubMed]
- Papadopoulos A, Porfyridis I, Christodoulides G, et al. A rare clinical case - Solitary fibrous tumor of the pleura. Respir Med Case Rep 2015;16:117-9. [Crossref] [PubMed]
- Tan J, Hsu A. Challenges in diagnosis and management of giant solitary fibrous tumour of pleura: a case report. BMC Pulm Med 2016;16:114. [Crossref] [PubMed]
- Anton C, Bojan V, Damjan V, et al. Giant solitary fibrous tumour of the pleura. Case report and review of the literature. Radiol Oncol 2015;49:395-401. [Crossref] [PubMed]
Cite this article as: Reig-Oussedik N, Rami-Porta R, Luizaga LA, Lloreta J, Serra-Mitjans M, Obiols C, Call S, Belda-Sanchis J. A patient with a localized malignant pleural mesothelioma and 15-year disease-free survival. Curr Chall Thorac Surg 2019;1:18.